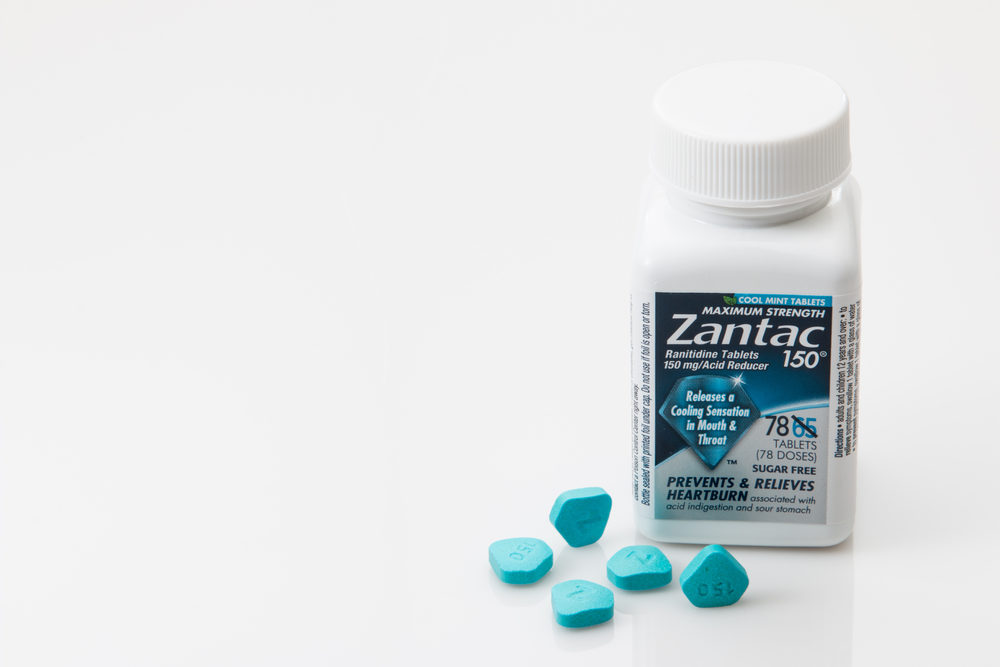
Generic Ranitidine Manufacturers Try to Get Class-Action Case Dismissed
On April 1, 2020, the U.S. Food and Drug Administration (FDA) requested the recall of all Zantac (ranitidine) antacid products from the market. Many of the products had tested positive for unsafe levels of N-nitroso dimethylamine (NDMA), a cancer-causing substance. Following that recall, many individuals…